The remarkable regenerative abilities observed in planarians and cnidarians are closely linked to the active proliferation of adult stem cells and the precise differentiation of their progeny, both of which typically deteriorate during aging in low regenerative animals. While regeneration-specific genes conserved in highly regenerative organisms may confer regenerative abilities and long-term maintenance of tissue homeostasis, it remains unclear whether introducing these regenerative genes into low regenerative animals can improve their regeneration and aging processes.
Here, we ectopically express highly regenerative species-specific JmjC domain-encoding genes (HRJDs) in Drosophila, a widely used low regenerative model organism. Surprisingly, HRJD expression impedes tissue regeneration in the developing wing disc but extends organismal lifespan when expressed in the intestinal stem cell lineages of the adult midgut under non-regenerative conditions. Notably, HRJDs enhance the proliferative activity of intestinal stem cells while maintaining their differentiation fidelity, ameliorating age-related decline in gut barrier functions.
These findings together suggest that the introduction of highly regenerative species-specific genes can improve stem cell functions and promote a healthy lifespan when expressed in aging animals.
Regeneration, an intricate process that rebuilds lost body parts, is a widespread phenomenon among metazoans, but the capacity for regeneration displays significant variation across different groups and species [1,2,3,4,5]. While certain animals like planarians and hydras possess the remarkable ability to regenerate their entire body from a small fragment, other groups with more complex body structures, such as mammals and insects, exhibit a diminished regenerative potential and can only regenerate specific tissues and/or organs to a limited extent. Furthermore, regenerative capacity often declines with aging in most species with limited regeneration abilities [2], resulting in increased susceptibility to organismal death upon injury. In contrast, animals that can achieve whole body regeneration, along with developmental reversion observed in the jellyfish Turritopsis, exhibit potential immortality [2, 5, 6]. Understanding the mechanisms underlying high regenerative ability and their relationship with aging represents a fundamental challenge in the field of developmental biology and gerontology with implications for regenerative medicine.
Several cellular and molecular factors have been identified as determinants of regeneration capacity. Highly regenerative animals such as planarians and cnidarian polyps rely on pluripotent adult stem cells, called neoblasts and interstitial cells (i-cells), respectively [2,3,4,5, 7]. These stem cells migrate to the injury sites and contribute to the formation of a blastema, an undifferentiated cellular mass, enabling the restoration of amputated body structures. Some vertebrates like salamanders and fish, which do not possess adult pluripotent stem cells, can regenerate organs after injury by recruiting blastema cells through dedifferentiation and/or the activation of quiescent lineage-restricted stem cells [1, 2, 4, 5, 8]. At the molecular level, the evolutionary conserved WNT signaling pathway promotes a wide range of regenerative events across species, including blastema formation in newts and Hydra [1,2,3,4,5, 8].
In contrast to the conserved regulators of regeneration, several genes are specific to highly regenerative animal groups and species: for instance, the newt gene Prod1 regulates re-patterning during limb regeneration [9, 10], and viropana family (viropana 1–5) is upregulated during lens regeneration [11, 12]. These species/group-specific genes might explain differences in regeneration capacity between species. Remarkably, ectopic expression of viropana 1–5 can enhance regeneration of the primordium of Drosophila eyes that maintain regenerative capacity during development [12]. This finding raises the possibility that heterologous induction of regenerative genes may accelerate tissue regeneration, at least in developing animals, and potentially provide a cue for developing novel regenerative therapies. However, it remains unknown whether heterologously-induced regenerative genes can improve regenerative and/or aging processes even when induced in post-developmental mature adults.
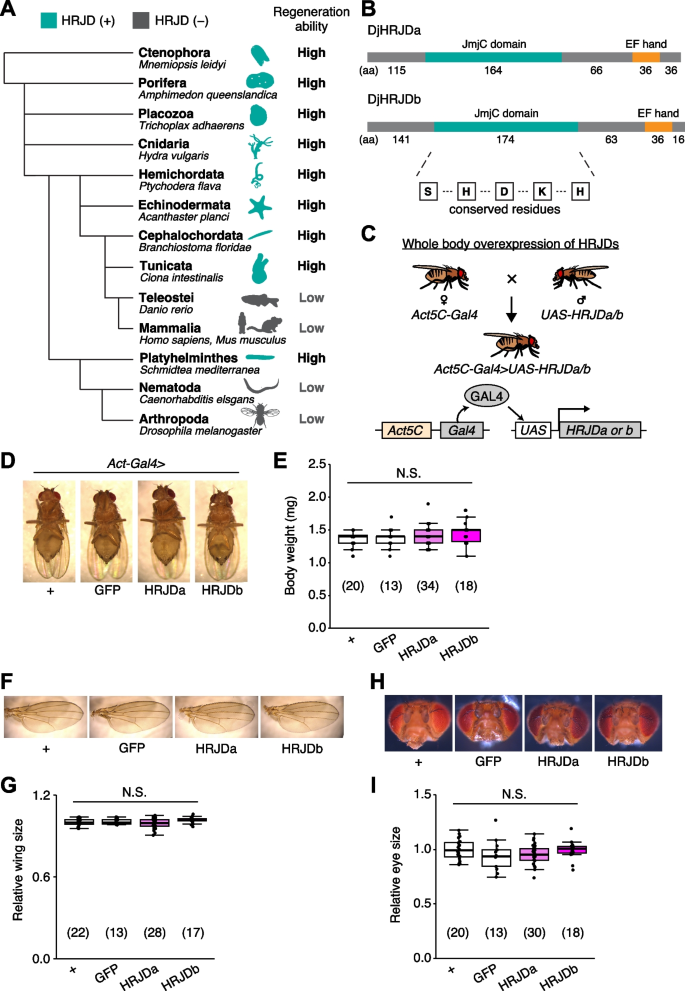
Notably, given that basal metazoans such as Porifera, Ctenophore, Placozoa, and Cnidaria all exhibit robust regenerative abilities, it is conceivable that a common ancestor of all metazoans once possessed a high regenerative potential and independently lost genes related to high regenerative capacity in multiple phyla. Building upon this hypothesis, bioinformatics analysis has identified genes that are common among species with high regenerative abilities and absent in species with limited regenerative capacities (Fig. 1A) [13]. The highly regenerative species-specific JmjC domain-encoding genes (HRJDs) are a group of such genes (with typically two or three orthologs per species) characterized by their JmjC domain (Fig. 1A), yet their molecular functions remain unknown. Given their potential influence on the regenerative process, HRJDs may contribute to the high regeneration potential of highly regenerative animals. With this in mind, a question arises: what would happen if a low regenerative species, which has lost HRJDs, were to acquire them again? By ectopically expressing HRJDs in low regenerative animal models, we can investigate their impacts on regeneration as well as on aging processes, providing insight into the role of HRJDs.

Here, we express HRJDs in the fruit fly Drosophila melanogaster and evaluate their impact in vivo, especially by focusing on two epithelial tissues: developing wing discs and post-developmental adult midguts, both of which exhibit regeneration potential and can replenish damaged epithelial cells. In contrast to the predicted contribution of HRJDs in regeneration as observed in planarian, ectopic HRJD induction impedes regenerative responses and decreases organismal survival upon injury in Drosophila. Surprisingly, however, HRJD expression in the stem/progenitor population of the adult midguts extends organismal lifespan under the non-regenerative condition. Further investigations reveal that HRJDs enhance the proliferative activity of intestinal stem cells while keeping their differentiation fidelity in aged guts, ameliorating age-related decline in gut barrier functions. These findings provide evidence that genes specific to highly-regenerative animals can improve stem cell function as well as increase healthy lifespan upon heterologous expression in aging animals.
Planarians are one of the most highly regenerative animals; they are capable of regenerating most body parts upon amputation and can even reconstruct their whole body from fragments [1,2,3,4, 8, 14]. Previous work has identified two HRJD orthologs HRJDa and HRJDb from two planarian species, Dugesia japonica and Schmidtea mediterranea, where both HRJDs contain only the JmjC domain and the EF hand motif (Fig. 1B) [13]. In functional assays using RNAi-mediated knockdown in D. japonica, these two HRJDs affect viability of organisms after amputation [13], suggesting that planarian HRJDs are associated with regeneration processes. We thus utilized these functional HRJDs in D. japonica as representatives for the following studies and hereafter simply named them as HRJDa and HRJDb.
To examine potential benefits and/or disadvantages of acquiring HRJDs, we then ectopically expressed HRJDs in Drosophila melanogaster, which has lost HRJD genes during evolution (Fig. 1A), using the Gal4/UAS system (Additional file 1: Fig. S1) [15]. We first introduced HRJDs in the whole body throughout development with the ubiquitous driver Act5C-Gal4 (Fig. 1C, Act5C-Gal4 > UAS-HRJDa/b). The whole body expression of HRJDs neither caused developmental lethality nor changed the body weight of mature adults compared with the Act5C-Gal4 > UAS-GFP control (Fig. 1D, E). We further assessed the gross morphology of adult wings and eyes under the ubiquitous expression of HRJDs. The wing size was not altered by HRJD expression (Fig. 1F, G). Similarly, Act5C-Gal4 > UAS-HRJDa/b flies did not change the size of adult compound eyes (Fig. 1H, I). These results indicate that HRJD expression does not disturb gross morphology of adult flies under homeostatic conditions, likely due to minimal impacts on developmental processes.
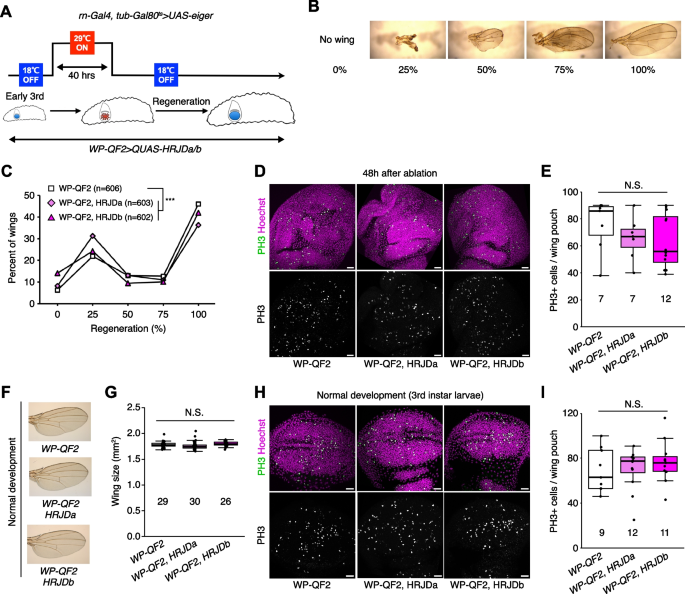
Given that HRJDs are conserved only among highly regenerative animals, their primary functions may be related to regeneration processes. Indeed, both planarian HRJDa and HRJDb function in whole-body regeneration while their relative contribution is likely context-dependent: HRJDa is indispensable for the regeneration of amputated heads while HRJDb promotes organismal survival after two consecutive amputations [13]. To test whether heterologously-induced HRJDs can enhance regenerative responses in Drosophila, we examined their impacts on regeneration after ablation of the developing wing imaginal disc. The Drosophila larval imaginal discs, including wing discs, which are composed of columnar epithelial cells, exhibit regenerative capacity and restore morphology even after massive cell death [16, 17] (Fig. 2A, B). We utilized the genetic ablation system in which transient overexpression of eiger, a Drosophila TNF ligand, induces apoptosis in the wing pouch region [17, 18]. In this tissue ablation system, the temperature-sensitive form of the Gal4 repressor Gal80 (tub-Gal80 ts ) allows transient tissue ablation under the control of a wing pouch driver rn-Gal4. To circumvent the temporal expression associated with the Gal4/Gal80 ts system, we further introduced an additional binary expression system: the QF/QUAS system for HRJD induction (Fig. 2A) [19, 20]. When expressing HRJDs in the wing pouch with the WP-QF2 driver during the entire process of recovery, we found that WP-QF2 > QUAS-HRJDs flies exhibit severe defects compared to controls (Fig. 2C). Immunostaining of a mitotic marker, phosphohistone H3 (PH3), revealed that HRJD expression did not cause statistically significant difference in regenerative proliferation in the wing pouch (Fig. 2D, E). Nevertheless, it is possible that insufficient damaged cell replenishment may occur in these conditions. Importantly, HRJD induction in the wing disc affected neither the development of the adult wing nor proliferation in wing discs under homeostatic condition (Fig. 2F–I). These results suggest that HRJD expression does not facilitate regeneration in the developing wing disc epithelium.
Most highly regenerative animals exhibit regeneration potential in mature adult stages [2,3,4, 8], and thus one possibility is that HRJDs stimulate regenerative responses only in adults, where tissue-resident stem cells play an important role in homeostasis and regeneration. We thus tested the impact of HRJD expression on the regenerative capacity of the Drosophila adult midgut. In the adult midgut, intestinal stem cells (ISCs) self-renew ISCs themselves and also generate differentiating progenitor cells called enteroblasts (EBs) and enteroendocrine progenitors (EEPs), which eventually differentiate into absorptive enterocytes (ECs) and secretory enteroendocrine cells (EEs), respectively (Fig. 3A) [21,22,23,24]. The adult midgut activates proliferation of ISCs in response to orally-treated harmful chemicals such as paraquat and dextran sulfate sodium (DSS), a regenerative response that is essential for organismal survival [21, 25,26,27,28]. We thus expressed HRJDs in ISCs and EBs using the esg-Gal4 (esg-Gal4 > UAS-HRJDa/b) driver and tested survival against paraquat/DSS damage. Similar to the wing regeneration assay, continuous expression of HRJDs via esg-Gal4 significantly impaired organismal survival upon the chemical challenges, with a stronger effect for HRJDa (Additional file 1: Fig. S2A and S2B). By contrast, survival during 11 days of control feeding (5% sucrose) was not significantly decreased by HRJD expression (Additional file 1: Fig. S2C), raising a possibility that abnormal regenerative response compromises the survival upon intestinal damage. We thus examined ISC proliferation after paraquat feeding by counting the number of cells marked with a mitotic marker, anti-PH3 staining, and found that induction of HRJDa significantly suppressed ISC division (Additional file 1: Fig. S2D), which is consistent with the stronger decline in survival rate. Concordant with the wing ablation experiments, these results suggest that continuous HRJD expression causes detrimental effects to regenerative capacity.
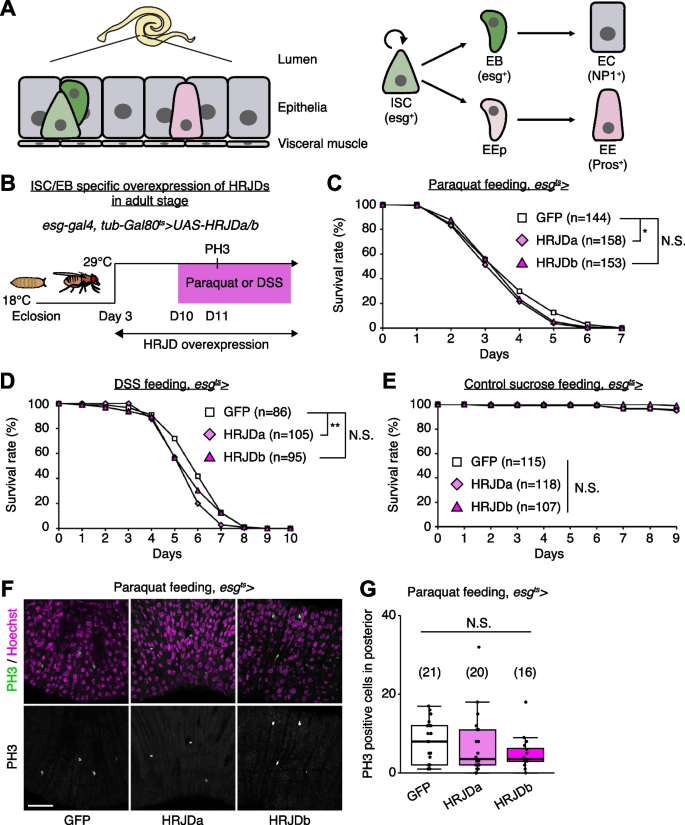
Because the esg-Gal4 driver is active not only in adult ISCs/EBs but also in the proliferative adult midgut progenitors in the larval midguts and larval imaginal discs [29, 30], continuous HRJD expression may cause internal organ defects associated with development. To temporally restrict HRJD expression to the adult stage, we next set up an experiment where HRJD expression starts only in adults after eclosion from pupa by combining esg-Gal4 with tub-Gal80 ts (esg-Gal4 ts > UAS-HRJDa/b) (Fig. 3B); however, neither HRJDa nor HRJDb improved organismal survival upon paraquat/DSS feeding (Fig. 3C, D). Instead, HRJDa expression caused a slight but significant decrease in survival rate compared to controls (esg-Gal4 ts > UAS-GFP), while HRJDb expression did not lead to a statistically significant decline of survival (Fig. 3C, D). Survival under control feeding (5% sucrose) was comparable between esg-Gal4 ts > UAS-GFP and esg-Gal4 ts > UAS-HRJDa/b (Fig. 3E), suggesting that the impaired survival of esg-Gal4 ts > UAS-HRJDa is specific to regenerative contexts. We also found that, after paraquat feeding, mitotic cell numbers in HRJD expressing flies decreased slightly (Fig. 3F, G), suggesting the possibility of impaired regeneration. These results indicate that post-developmental expression of HRJDs does not enhance intestinal regeneration in adult flies.
Highly-regenerative animals are often resistant to organismal aging and have long lifespan; some species like the jellyfish Turritopsis even revert their early developmental stages under harsh environments and are considered to be potentially immortal [2, 6]. Although heterologous expression of HRJDs does not augment tissue regeneration in either developing or adult Drosophila tissues, we further investigated whether HRJDs influence organismal lifespan under homeostatic conditions in which no experimental injuries are applied to fly adults. We observed that post-developmental HRJD expression in adult ISCs/EBs (esg-Gal4 ts > UAS-HRJDa/b) significantly extended organismal lifespan both in females (Fig. 4A) and males (Fig. 4B). In contrast, continuous HRJD expression in the whole body (Act5C-Gal4 > UAS-HRJDa/b) shortened males’ lifespan (Fig. 4C, D). Moreover, HRJD expression in differentiated ECs (NP1-Gal4 > UAS-HRJDa/b) also negatively impacted female lifespan, likely due to developmental abnormalities since EC-specific HRJD expression in the adult stage did not decrease survival (Additional file 1: Fig. S3A-S3D). These results suggest that spatio-temporally regulated induction of HRJDs in adult ISCs/EBs can be beneficial for adult flies under homeostatic conditions.
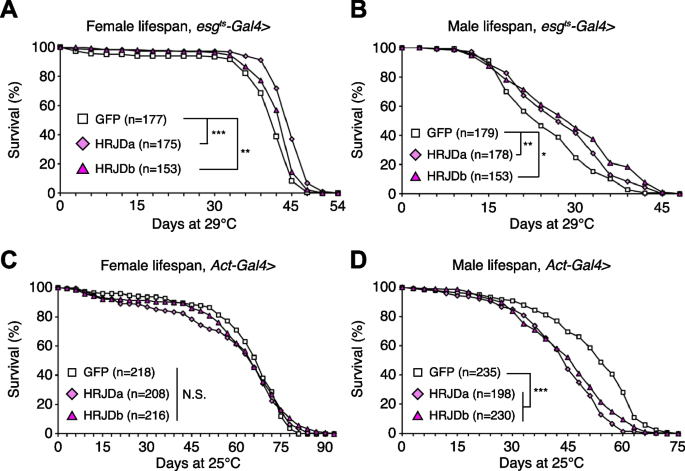
Further investigation revealed the distinct impacts of HRJDa and HRJDb on organismal lifespan: while continuous expression of HRJDa via esg-Gal4 (esg-Gal4 > UAS-HRJDa) shortened lifespans for both males and females, esg-Gal4 > UAS-HRJDb prolonged female lifespans but shortened male lifespans (Additional file 1: Fig. S3E and S3F). The ortholog-dependent phenotypic differences were also reported in planarian regeneration [13]. To address the mechanisms of lifespan extension associated with post-developmental HRJD expression in ISCs/EBs, we focused on esg-Gal4 ts > UAS-HRJDs midguts in the following experiments.
Age-related mortality accompanies intestinal barrier dysfunction and disruption of cell–cell junction in the gut epithelium across species [31,32,33,34]. Given that aging phenotypes are more severe in female flies than male flies [35], we first tested barrier function of the esg-Gal4 ts > UAS-HRJDa/b female midgut using the Smurf assay, in which orally-administered blue dye spreads throughout the entire body when the intestinal barrier is disrupted (Smurf + , Fig. 5A) [31, 35]. Consistent with lifespan extension, post-developmental HRJD expression in ISCs/EBs significantly decreased the ratio of Smurf + adults at 30 days old, a time point where age-related organismal death starts (Fig. 4A and Fig. 5A), suggesting that barrier function is maintained in aged flies that express HRJDs in ISCs/EBs. We further assessed the localization of septate junction markers (Dlg, Tsp2A, Mesh, Ssk), which diffuse in cytoplasm in the aged midgut [33, 36,37,38]. The diffusion of the Dlg protein in aged midguts was significantly suppressed in esg-Gal4 ts > UAS-HRJDa/b midguts (Fig. 5B, C). Similarly, peripheral localization of Tsp2A, Mesh, and Ssk proteins was also maintained in esg-Gal4 ts > UAS-HRJDa/b midguts compared with control (esg-Gal4 ts > UAS-GFP) midguts (Fig. 5D–I). Given that HRJDs in ISCs/EBs affect localization of junctional components in ECs, we next tested if HRJDs non-autonomously affect junctional integrity. To this end, we performed a mosaic experiment where HRJDs were clonally induced using the esgFLPout system [26]. Clonal induction of HRJDs improved junctional localization of Ssk both inside and outside the clones (Additional file 1: Fig. S4A and S4B), supporting the conclusion that HRJDs’ effect is non-cell autonomous. We then examined JNK activation in ECs, which is associated with barrier dysfunction in the aged intestine [27, 36]. HRJD expression in ISCs/EBs significantly repressed transcription of puckered (puc), a downstream target of JNK signaling in the whole midgut (Fig. 5J), and indeed the intensity of puc-lacZ reporter decreased in ECs upon HRJD induction (Fig. 5K, L). HRJD expression in ISCs/EBs thus suppresses JNK signaling in ECs in the aged midgut, which is mediated by either a non-autonomous function of HRJDs or residual HRJD proteins in newly differentiated ECs. These results indicate that HRJD expression in adult ISCs/EBs contributes to the prolonged maintenance of the junctional integrity as well as gut barrier function in the aged intestine.
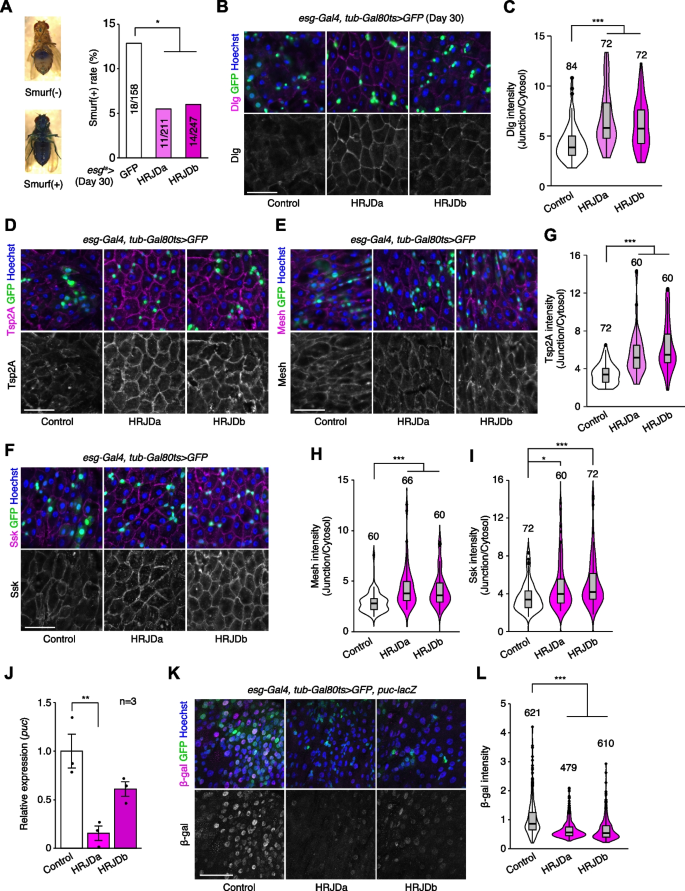
In addition to the intestinal barrier dysfunction, aberrant activation of ISC mitosis and accumulation of mis-differentiated ISC progenies are also common age-related phenotypes in the Drosophila adult midgut [27, 31, 33, 39]. In 30-days old control midguts, esg-Gal4 ts > UAS-GFP + cells exhibited hallmarks of age-related mis-differentiation into ECs such as increased ploidy and enlarged nuclear size (Fig. 6A) [27]. By contrast, we found that HRJD expression in adult ISCs/EBs suppressed these mis-differentiation phenotypes (Fig. 6A, B), which are consistent with the amelioration of age-related barrier dysfunction in esg-Gal4 ts > UAS-HRJDa/b midguts. Surprisingly, however, esg-Gal4 ts > UAS-HRJDa/b did not suppress age-related increase in ISC proliferation but rather enhanced it in 30-day old midguts (Fig. 6C, D). The enhancement of ISC proliferation was specific to the aged intestine since esg-Gal4 ts > UAS-HRJDs did not affect PH3 + cell number in 10-day-old young midguts (Additional file 1: Fig. S4E). These results raised the possibility that HRJD expression augments the mitotic activity of stem cells while maintaining their differentiation fidelity during aging.
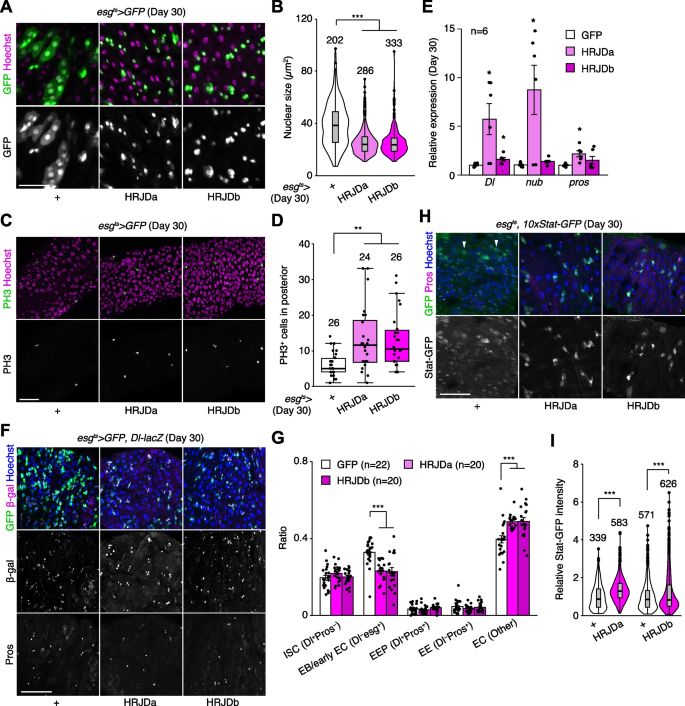
To test this hypothesis, we first measured the expression of cell type markers (Delta for ISCs, nub for ECs, and pros for EEs) by RT-qPCR of whole midguts. Both nub and pros are negatively regulated by Esg [40, 41], and accumulation of esg + mis-differentiated cells accompanies the downregulation of EC-related genes in aged midguts [27]. Consistent with enhanced ISC proliferation, esg-Gal4 ts > UAS-HRJDa significantly upregulated Delta (Dl) expression in 30-day old midguts (Fig. 6E). More importantly, esg-Gal4 ts > UAS-HRJDa also upregulated nub and pros, which supports our hypothesis that HRJDs improve proper differentiation of ISC progenies. In esg-Gal4 ts > UAS-HRJDb, only Dl, but not nub and pros, was upregulated when whole midguts were used as sample (Fig. 6E). Next, we focused on the posterior midgut where both HRJDa and HRJDb prevent mis-differentiation of EBs (Fig. 6A) and examined cell type composition using the combination of markers esg ts > GFP (ISC/EB), Dl-lacZ (ISC/EEP), and anti-Pros (EEP/EE). Consistent with the prevention of EB mis-differentiation, esg ts -mediated HRJD expression decreased EBs (esg + Dl − Pros − ) and increased polyploid ECs (esg − Dl − Pros − ) compared to the control midgut (Fig. 6F, G). These results indicate that HRJDs improve differentiation fidelity in ISC lineage.
To address the potential mechanism of HRJD-dependent maintenance of ISC functions, we focused on the JAK-STAT pathway since its activation promotes both ISC proliferation and differentiation into ECs [26, 42, 43]. In young mature midguts, STAT activity is largely restricted to esg + ISCs/EBs [26, 42, 44]. In aged midguts, however, a subset of polyploid cells exhibited weak signal of the STAT reporter 10 × STAT-GFP, likely due to the accumulation of mis-differentiated EBs (Fig. 6H). Notably, HRJD induction canceled such ectopic STAT activation in polyploid cells and rather enhanced STAT activity in diploid ISCs/EBs (Fig. 6H, I), which is consistent with our observations that HRJDs boost ISC functions during aging. These results suggest that HRJD expression accelerates the generation of differentiated ECs via JAK-STAT activation. In the healthy homeostatic intestine, ISC division and subsequent differentiation into ECs is coupled with the loss of old ECs [45, 46]. Interestingly, we found that esg-lineage clones that express HRJDs rapidly replaced pre-existing cells by generating polyploid ECs (Additional file 1: Fig. S4A, S4C, and S4D). In addition, more cells exhibited cleaved Dcp1, an apoptotic marker [47, 48], in 30-day old esg ts > HRJDs midguts (Additional file 1: Fig. S4F and S4G), implying that HRJD induction promotes turnover of midgut epithelial cells. Collectively, our data show that post-developmental expression of HRJDs improves stem cell functions in the aged intestine.
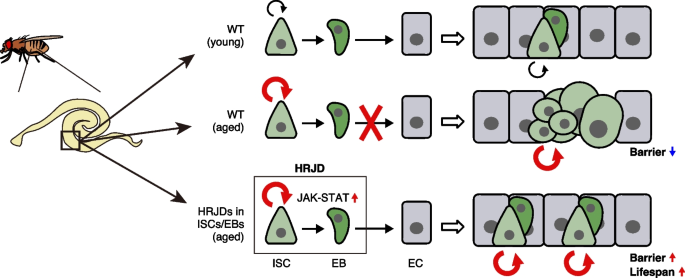
In this study, we demonstrate that post-developmental expression of planarian HRJD genes in the Drosophila adult ISCs/EBs can suppress age-related intestinal dysfunctions and extend organismal lifespan, while their continuous expression throughout the entire developmental process hampers regenerative responses and principally shortens lifespan. Notably, HRJDs in adult ISCs/EBs boost the age-related increase of ISC proliferation but do not cause age-related mis-differentiation of ISC progenies (Fig. 7). These HRJD-mediated outcomes are distinct from those mediated by typical anti-aging manipulations such as antibiotic treatment and metabolic intervention, which ameliorate both ISC over-proliferation and mis-differentiation [24, 31, 49]. Therefore, heterologously induced HRJDs create the neomorphic state in the aged intestine (Fig. 7). Given the age-related increase of chronic cellular stresses [24, 27, 36, 49, 50], we speculate that the HRJD-dependent ISC activation and their proper differentiation improve organismal fitness by enabling active turnover of damaged intestinal cells.
Our investigation implies that the activation of the JAK-STAT pathway underlies the HRJD-mediated enhancement of ISC functions. Surprisingly, however, the expression of most mitogens including upstream ligands of JAK-STAT signaling (upd1, upd2, upd3) were not upregulated by HRJD induction (Additional file 1: Fig. S4H), raising the possibility that HRJDs activate STAT by modulating intracellular signaling factors. Such ligand-independent STAT activation can be achieved via non-receptor type tyrosine kinases like Src and Abl [51]. Given that the typical JmjC family proteins function as histone demethylase or protein hydroxylase [52], HRJDs may epigenetically target these tyrosine kinases. Notably, Drosophila Jarid2 (Jumonji, AT rich interactive domain 2) activates EGFR signaling upon overexpression in ISCs/EBs without changing ligand expression [53]. Our findings, together with these reports, suggest a cell-autonomous role of JmjC family proteins in regulating proliferative signaling in stem cell lineages. Intriguingly, Jarid2 expression in ISCs/EBs leads to ISC over-proliferation as well as barrier dysfunction, resulting in reduced lifespan [53], which is the opposite adult phenotype of HRJD expression in ISC lineages. Moreover, in contrast to the nuclear localization of histone demethylase KDM8 [54, 55], the closest paralog of HRJDs, ectopically induced HRJDs localize in the cytoplasm but not in the nucleus both in vitro (S2 cells) and in vivo (wing discs and adult midguts) (Additional file 1: Fig. S1B-S1D). This is a common localization pattern of JmjC-domain only proteins [55, 56] and implies that an unknown mechanism may operate when HRJDs work as a potential histone demethylase. In the future, it will thus be critical to examine the detailed molecular function of HRJDs to understand their impact on stem cell lineages.
Although HRJDs have been identified as genes conserved between highly regenerative animals [13], it is unclear whether HRJDs alone are sufficient to enhance regenerative responses. Our investigations revealed that ectopic expression of HRJDs failed to improve regeneration of developing wing discs and adult midguts in Drosophila (Figs. 2 and 3), likely through the attenuation of regenerative growth. However, it should be noted that we induced HRJDs before tissue injury and maintained their expression during regeneration period. Given that HRJDb is upregulated in the late phase of planarian regeneration (3 days after amputation) [13], strict regulation of HRJD induction might be important for their proper function as true regeneration regulators. On the other hand, the expression level of HRJDa remains constant during planarian regeneration [13], which is recapitulated in our experiments. Another possibility is that a high regeneration ability can be achieved by cooperation between HRJDs and other unidentified genes that have been lost during evolution, which should be addressed in future studies.
During the normal aging process in the Drosophila adult midgut, ISC over-proliferation is closely linked with other aging phenotypes, and suppression of ISC division can prevent the accumulation of mis-differentiated ISC progenies and extend organismal lifespan [24, 36, 49, 50]. In contrast, HRJDs enhance age-related ISC activation but can alleviate mis-differentiation and extend lifespan, suggesting that it is not ISC activation itself but rather mis-differentiation of ISC progenies that principally drives age-related mortality. Consistently, manipulation of mitotic spindle orientation, which affects the cell fate of daughter cells [57,58,59], can extend organismal lifespan without changing mitotic activity of ISCs [59]. Of note, in contrast to Drosophila ISCs which over-proliferate in aged midguts, many adult stem cells decrease their activity and abundance during aging both in Drosophila and mammals [24, 60, 61]. Future investigations for mechanisms of HRJD-mediated stem cell rejuvenation will provide clues to develop new anti-aging strategies.
In this study, we established a Drosophila model in which HRJDs are heterologously expressed in specific tissue or cell type using binary expression systems and demonstrated that HRJDs can improve proliferative/differentiation capacity of ISCs in the aged midgut. Although continuous HRJD expression in the wing disc and in the adult midgut impairs tissue regeneration upon injury, restricted HRJD expression in post-developmental adult ISCs/EBs enhances mitotic activity of ISCs as well as maintains their differentiation fidelity in the aged flies, leading to the prevention of age-related intestinal barrier dysfunction and the extension of organismal lifespan. Our HRJD-expressing model will serve as a valuable resource to understand unprecedented mechanisms of stem cell rejuvenation in the future.
All stocks were maintained on a standard diet containing 4% cornmeal, 6% baker’s yeast (Saf Yeast), 6% glucose (Wako, 049–31177), and 0.8% agar (Kishida chemical, 260–01705) with 0.3% propionic acid (Tokyo Chemical Industry, P0500) and 0.05% nipagin (Wako, 132–02635). Canton S was utilized as the wildtype strain. Transgenic fly lines were obtained from the Bloomington Drosophila Stock Center (BDSC, https://bdsc.indiana.edu/) and the Kyoto Stock Center (https://kyotofly.kit.jp/cgi-bin/stocks/index.cgi). Unless otherwise indicated, strain descriptions can be found at Flybase (http://flybase.bio.indiana.edu): Act5C-Gal4 (BDSC 3954, described in Flybase and BDSC), rn-Gal4, tub-Gal80ts, UAS-egr [17] (BDSC 51280), WP-QF2 [62], tub-Gal80ts (BDSC 7017, 7019, described in Flybase and BDSC), esg-Gal4 [63] (Kyoto 109,126), NP1-Gal4 [64] (Kyoto 112,001), UAS-GFP (BDSC 1521, described in Flybase and BDSC), MZH-86Fb [65] (Kyoto 130,437), UAS-HRJDa (this study), UAS-HRJDb (this study), QUAS-HRJDa (this study), QUAS-HRJDb (this study), UAS-FLAG-HRJDa (this study), UAS-FLAG-HRJDb (this study), puc-lacZ [66] (BDSC 98329), 10 × STAT-GFP [67] (BDSC 26198), Dl-lacZ [68] (BDSC 11651). esg-Gal4, UAS-GFP, tub-Gal80ts; UAS-FLP, Act5C-FRT.CD2-Gal4 (esgFLPout) [26, 69] is a gift from Irene Miguel-Aliaga. See Additional file 2: Table S1 for genotypes in each figure. We used female flies unless otherwise noted in the figures.
Experimental crosses that did not involve Gal80 ts -mediated inhibition of Gal4 were performed at 25 °C. When using Gal80 ts , experimental crosses were maintained at 18 °C. For genetic wing ablation experiments (Fig. 2), F1 larvae were raised at 18 °C until day 7, incubated at 29 °C for the next 40 h, and then maintained at 18 °C until the adult hatched or dissection. For lifespan assays, midgut staining, and Smurf assays (Figs. 3, 4, 5, and 6), F1 adults were transferred to 29 °C 3 days after eclosion until the experiments. Midgut staining and Smurf assays were performed after 7 or 27 days of 29 °C incubation (young: day 10 adults, old: day 30 adults). For esgFLPout experiments, flies were maintained at 18 °C until 50 days, and 50-day old adults were transferred to 29 °C and analyzed after 10 days (final 60-day old).
To express HRJDs using the Gal4/UAS or the QF/QUAS system, we constructed a vector containing HRJDa or HRJDb under the control of either the UAS sequence or QUAS sequence. Namely, we amplified HRJDa and HRJDb sequence from codon-optimized synthesized DNAs (pUCIDT-HRJDa and pUCIDT-HRJDb, IDT, Additional file 3: Supplementary texts S1) for DjHRJDa (Genbank # LC408963) and DjHRJDb (Genbank # LC408964), respectively. UAS-HRJDa and UAS-HRJDb were constructed by ligating HRJDa and HRJDb into pUAST-attB (DGRC 1419), respectively (digested by EcoRI/NotI). Similarly, QUAS-HRJDa and QUAS-HRJDb were constructed by ligating HRJDa and HRJDb into pQUAS-WALIUM20 (DGRC 1474), respectively (digested by NheI/EcoRI). To construct UAS-3xFLAG-HRJDs, we PCR-amplified the coding sequences of HRJDs from pUCIDT-HRJDs by gene-specific primers with 3xFLAG tag at 5′-end of the forward primers, and the amplicons were then ligated into a pUAST-attB vector digested by EcoRI/NotI using In-Fusion HD Cloning Kit (Takara, 639,649). The landing site for each construct was ZH-86Fb. Injection and selection were performed by WellGenetics (Taiwan, R.O.C.). Please see also Table S2 and Supplementary texts S1 for primer sequences and HRJDs sequences.
Drosophila S2 cells were grown at 25 °C in Schneider’s Drosophila medium (GIBCO, 21,720,001) supplemented with 10% (v/v) heat-inactivated fetal bovine serum (FBS), 100 U/mL penicillin, and 100 μg/mL streptomycin (FUJIFILM Wako, 168–23,191).
To examine the expression pattern of HRJDs through immunostaining, 1 × 10 6 cells were seeded in 6-well plates containing Schneider’s Drosophila medium supplemented with 10% (v/v) heat-inactivated fetal bovine serum, 100 U/mL penicillin, and 100 μg/mL streptomycin and were transfected with 800 ng of pAc5-3xFLAG-HRJDs using Effectene Transfection Reagent (QIAGEN, 301,427) following the manufacturer’s protocol. To prepare pAc5-3xFLAG-HRJDs, we amplified the coding sequences of HRJDs from pUCIDT-HRJDs (Additional file 3: Supplementary texts S1) by gene-specific primers with 3xFLAG tag at 5′-end of the forward primers, and the amplicons were then ligated into a pAc5-STABLE2-neo (Addgene, 32,426) [70] digested with EcoRI/XhoI by In-Fusion HD Cloning Kit (Takara, 639,649). Twenty-four hours after transfection, the cells were washed with PBS and the medium was replaced with fresh Schneider’s Drosophila medium supplemented with 10% (v/v) heat-inactivated FBS, 100 U/mL penicillin, and 100 μg/mL streptomycin. Forty-eight hours after transfection, the cells were fixed with 4% paraformaldehyde (PFA) in PBS, washed with PBS containing 0.1% Triton X-100 (PBST), blocked in PBST with 5% normal donkey serum (PBSTn), and incubated with primary antibodies in PBSTn overnight at 4 °C. The samples were then washed with PBST, incubated for 1 h at room temperature with secondary antibodies and Hoechst 33,342 suspended in PBSTn and washed again with PBST. Images were captured using an LSM880 (Zeiss). The primary antibody used was a mouse anti-FLAG M2 monoclonal antibody (1: 2000, Sigma, F1804). The secondary antibodies used were Goat anti-Mouse IgG2b Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 555 (1:2000, Thermo Fisher Scientific, A-21147). Hoechst 33,342 (0.4 μM; Invitrogen, H3570) was used for nuclear staining. Images were analyzed and edited using Fiji/ImageJ software (NIH).
To examine the expression of HRJDs, 2.5 × 10 5 cells were seeded in 24-well plates containing Schneider’s Drosophila medium supplemented with 10% (v/v) heat-inactivated FBS, 100 U/mL penicillin, and 100 μg/mL streptomycin and were transfected with 200 ng of pAc5-3xFLAG-HRJDs using Effectene Transfection Reagent (QIAGEN, 301,427) following the manufacturer’s protocol. Twenty-four hours after transfection, the cells were washed with PBS, and the medium was replaced with fresh Schneider’s Drosophila medium supplemented with 10% (v/v) heat-inactivated FBS, 100 U/mL penicillin, and 100 μg/mL streptomycin. Forty-eight hours after transfection, the cells were washed with PBS and lysed with 50 μL RIPA buffer supplemented with cOmplete ULTRA EDTA-free protease inhibitor cocktail (Roche, 05892953001). The lysate was sonicated and centrifuged at 20,000 g for 5 min. Protein concentration was determined by BCA assay. The supernatant was mixed with SDS, boiled at 95 °C for 5 min, and subjected to western blotting.
Proteins were separated using SDS-PAGE and transferred onto Immobilon-P PVDF membranes (Millipore, IPVH00010) for immunoblotting. Membranes were blocked with 4% skimmed milk diluted in 1 × Tris buffered saline containing 0.1% Tween-20. Immunoblotting was performed using the below-mentioned antibodies, which were diluted with 4% skim milk. The signals were visualized using Immobilon Western Chemiluminescent HRP Substrate (Millipore, WBKLS0500) and FUSION SOLO. 7S. EDGE (Vilber-Lourmat). Contrast and brightness adjustments were applied using the Fiji/ImageJ software (NIH).
The primary antibody used was mouse anti-FLAG M2 monoclonal antibody (1: 5000, Sigma, F1804). Mouse anti-alpha tubulin (DM1A) monoclonal antibody (1: 5000, Sigma, T9026) was used as loading control. The secondary antibody used was HRP-conjugated goat/rabbit/donkey anti-mouse IgG (1: 10,000, Promega, W402B).
5 mM paraquat (Sigma, 856,177) and 5% (w/v) DSS (MP Biomedicals, 160,110) were dissolved in 5% (w/v) sucrose solution. Filter paper (Whatman 3MM) was soaked with 400 μL of these reagents and placed into empty vials. For histological analyses, flies were fed with the reagent solution for 1 day. For the survival assay, flies were transferred to new vials and dead flies (determined by immobility and showing no response to tapping) were counted every day. 5% sucrose was used for control feeding.
Samples were dissected in 1 × PBS and fixed in 4% PFA for 20 min (wing discs) and 30–45 min (adult midgut) at room temperature (RT), respectively. The following primary antibodies were used with indicated dilution into 1 × PBS containing 0.5% BSA and 0.1% Triton X-100: rabbit anti-PH3 (Millipore 06–570, 1:1000), mouse anti-FLAG (Sigma F1804, 1:1000), mouse anti-Dlg (DSHB 4F3, 1:100), rabbit anti-GFP (MBL 598, 1:500), rat anti-GFP (Nacalai tesque 04404–26, 1:400), rabbit anti-Tsp2A (Izumi et al., 2016, 1:1000) [71], rabbit anti-Mesh (Izumi et al., 2012, 1:1000) [72], rabbit anti-Ssk (Izumi et al., 2016, 1:1000) [71], chicken anti-β-galactosidase (Abcam ab9361, 1:500), anti-cDcp1 (Cell Signaling Technology 9578, 1:200). After overnight incubation with primary antibodies at 4 °C, samples were incubated with fluorescent secondary antibodies (Jackson ImmunoResearch and Invitrogen, 1:500) for 1 h at RT. Hoechst 33,342 (Invitrogen, final concentration: 10 μg/ml) was used to visualize DNA. Wing discs were mounted as described previously [73, 74]. Samples were mounted in Slowfade Diamond (ThermoFisher, S36963) and imaged with confocal microscopy Zeiss LSM880 or Zeiss LSM980.
We referred to Rera et al. (2012) for the Smurf assay conditions [35]. To prepare the feeding medium, 100 μL of 50% (w/v) brilliant blue FCF (Wako, 027–12842, final 2.5%) and 100 μL of 5% (w/v) sucrose were added to a vial containing 2 ml of cornmeal-agar food. After mixing with a spatula, a Whatman 1 filter (1001–020) was put on the feeding medium. Flies were fed with this medium at 25 °C for 1 day, after which they were transferred to a new vial containing cornmeal-agar food without blue dye to clean the epidermis. Two hours after the transfer, Smurf phenotype was checked. We classified the Smurf phenotype as blue dye leakage outside abdomen (thorax, head, legs).
Total RNA was purified from 10 to 15 midguts using the ReliaPrep RNA Tissue Miniprep System (Promega). cDNA was made from 100 or 200 ng of RNA using PrimeScript RT Reagent Kit (TaKaRa). Quantitative PCR was performed using TB Green Premix Ex Taq II (TaKaRa) and the QuantStudio 6 Flex Real-Time PCR System (ThermoFisher). RpL32 was used as an internal control. Primer sequences are listed in Additional file 4: Table S2.
We created the phylogenetic tree of representative organisms using the NCBI taxonomy browser (https://www.ncbi.nlm.nih.gov/Taxonomy/CommonTree/wwwcmt.cgi). The species examined are Amphimedon queenslandica (Porifera), Mnemiopsis leidyi (Ctenophora), Trichoplax adhaerens (Placozoa), Hydra vulgaris (Cnidaria), Ptychodera flava (Hemichordata), Acanthaster planci (Echinodermata), Branchiostoma floridae (Cephalochordata), Ciona intestinalis (Tunicata), Danio rerio (Teleostei), Homo sapiens, Mus musculus (Mammalia), Schmidtea mediterranea (Platyhelminthes), Caenorhabditis elegans (Nematoda), and Drosophila melanogaster (Arthropoda). These species, except for D. rerio, H. sapiens, M. musculus, C. elegans, and D. melanogaster, are known to have HRJDs and exhibit high regeneration ability [13]. The pictures were downloaded from PhyloPic, and the color of some were changed from black to blue. Credits: Bennet McComish (B. floridae), Malio Kodis (M. leidyi), and Markus A. Grohme (S. mediterranea, https://www.phylopic.org/images/93f9611c-0cbd-4a14-92da-004a4521e21f/schmidtea-mediterranea).
For wing discs, we counted PH3 positive cells in the pouch region, which develops into the adult wing. After binarization, the number of PH3 positive cells was counted using the Analyze Particles function in Fiji/ImageJ and subsequently confirmed or corrected by visual review of the images. For adult midguts, the number of PH3 positive cells in the posterior midguts was manually counted.
To quantify junctional and cytoplasmic intensity, a line that was orthogonal to one side of a cell was drawn, and then the plot profile was calculated for the line. We defined the junctional intensity as the highest value in that profile and defined cytoplasmic intensity as the value 20 pixels (~ 2.6 μm) away from the highest value. Three lines were drawn for one cell and the average of the three was used to represent the junctional/cytoplasmic ratio for the cell. Six polyploid cells were randomly selected from each image for quantification.
The signal intensity of puc-lacZ and 10 × STAT-GFP reporters was quantified using Fiji/ImageJ. The nuclei of large polyploid cells (for puc-lacZ) and Pros − diploid cells (for 10 × STAT-GFP) were selected as ROIs using the polygon selection tool. The reporter intensity in each ROI was quantified using the Measure command.
Nuclei of esg-Gal4 > UAS-GFP positive cells were traced by the polygon selection tool and added as ROI in Fiji/ImageJ. The size of each ROI was quantified using the Measure command.
Cell number was quantified using Fiji/ImageJ. To count the total cell number, Hoechst signal was processed as following: (1) despeckle, (2) binarization, (3) fill hole, (4) watershed, (5) analyze particle. The following cell number was manually counted using the cell counter function: esg > GFP + cells, Dl-lacZ + cells, Pros + cells, and cDcp1 + cells.
Statistical analyses were performed using Excel and RStudio. Two tailed t tests were used for comparisons between two groups. One-way ANOVAs with post hoc Tukey tests were performed when comparing three or more groups. Log-rank tests were used for comparison of survival curve. Significance is indicated in the figures as follows: *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001, not significant (N.S.): P > 0.05. Bar graphs show mean ± standard error. Boxplots show median (thick line in the box), first and third quartiles (bottom and top of the box), minimum value (lower whisker), and maximum value (upper whisker). Dots in bar graphs and boxplots indicate individual values. Violin plots portray the distribution of individual values.
All data generated or analyzed during this study are included in this published article and its supplementary information files. Drosophila stocks used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Highly regenerative species-specific JmjC domain-encoding gene